The placebo paradox: Why a COVID-19 vaccine trial participant might drop out
By Dawn Sinclair Shapiro | December 9, 2020
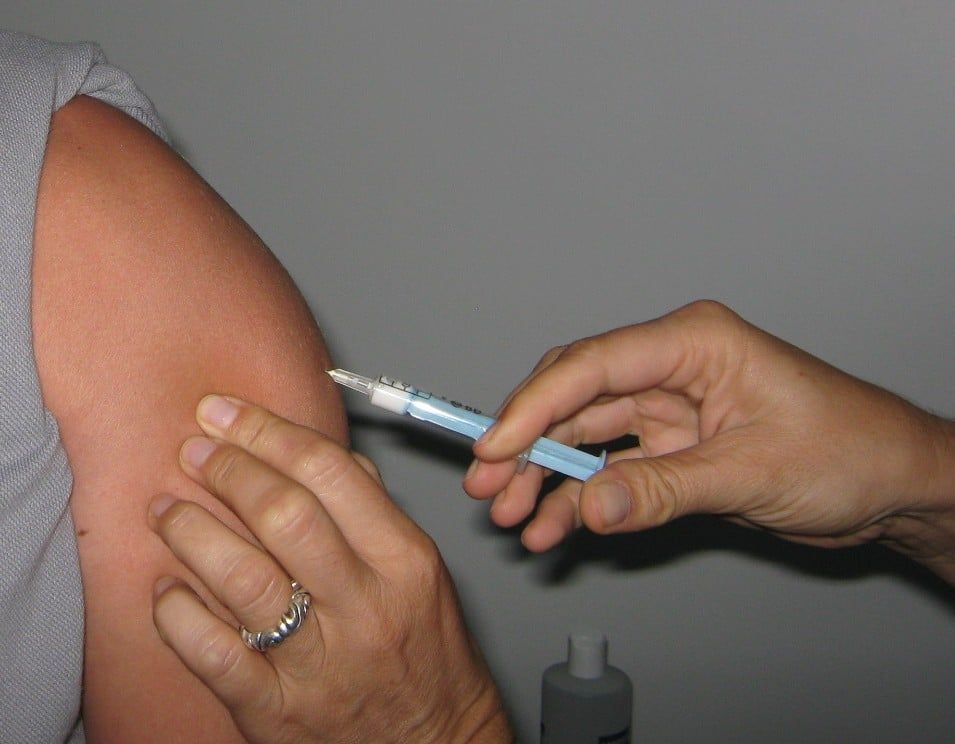
I’m a covid killer, a foot soldier in the global challenge to test life-preserving vaccines that will help end the pandemic. In September I signed up for Moderna’s two-year randomized control trial for its mRNA-1273 vaccine, which gave me a 50 percent chance that my jab contained an actual vaccine. So why might I soon quit the trial?
Family and friends initially met my foray into experimental vaccine research with concern and skepticism, but as a chronic asthma sufferer, I felt compelled to join the fight and potentially gain early vaccination. In general, I respect, yet scrutinize, the regulatory process and take immunization guidance from my physicians and public health experts. As a journalist, I’ve seen what wild polio does to humans when it attacks the nervous system and documented the 2009 vaccination campaigns in Northern Nigeria that helped eradicate the virus there. I’ve interviewed women rendered infertile from an intrauterine birth control device called the Dalkon Shield, a medical disaster that provoked lawsuits and led to the 1976 legislation that authorizes the US Food and Drug Administration (FDA) to regulate medical devices.
For more than a hundred years, randomized control trials—blinded studies in which volunteers are randomly injected with an experimental vaccine or assigned to a placebo control “arm” of the study—have represented the gold standard for determining the effectiveness of experimental medicines. By comparing the outcomes of the control (placebo) and active (vaccine) arms over a predetermined time period that often lasts for years, scientists and regulators determine if a new intervention actually works. Participants in such trials maintain the right to abandon a study at any time, at which point they are “un-blinded” and told whether they got a vaccine or a placebo. The COVID-19 crisis marks the first time the FDA has sought to accelerate a randomized control vaccine trial by using its protective authority to issue an emergency use authorization for a novel vaccine. The framework originated in biodefense under President George W. Bush and expanded in 2013 under President Barack Obama to “cover emerging infectious diseases.”
Assuming Pfizer and Moderna receive emergency use authorizations for their COVID-19 vaccines from the FDA as expected in the coming weeks, the vaccine data story will begin its next chapter, as millions of people begin receiving vaccines. The astonishing and rapid success of these vaccines—each was developed in less than year, and both are reported to be more than 90 percent effective—brings me to a crossroads. Not knowing which arm of the study I inhabit, should I abandon the study when my regular health care provider offers me a vaccine? Tens of thousands of trial participants could reluctantly choose personal certainty and leave their trials when vaccines become more widely available. With our departure, scientists and researchers will lose the long-term safety, efficacy, and durability data they require from a randomized control trial. Researchers may not be able to detect negative consequences that are slow to develop or to determine how long the immunity conferred by the vaccine lasts.
The dilemma of how to treat COVID-19 vaccine trial participants, once emergency use authorizations are granted, potentially pits vaccine makers against regulators. Pfizer and Moderna—which require continued participation in their trials to determine the long-term durability of their vaccines—believe the best method to preserve participation is to un-blind their studies and vaccinate their control arm participants. Earlier this week I received a reassuring email from the Moderna study explaining the emergency use authorization timeline and their intent to “ensure that all participants ultimately have the option of receiving the vaccine.”
Other vaccine makers, whose trials have yet to begin or are incomplete, face concerns about attracting and maintaining participants for their trials if alternatively approved vaccines are already available. Conversely, on Wednesday the World Health Organization’s AD Hoc Expert Group published its perspective on the placebo paradox in the New England Journal of Medicine, stating “we believe it is ethically appropriate to continue blinded follow-up of placebo recipients.” The FDA is internally deliberating the issue. Anthony S. Fauci, the director of the National Institute of Allergy and Infectious Disease, applied a moral rationale when speaking to a USA Today panel: “What you don’t want to do is having people on the outside readily having vaccine available to them, and you’re still having somebody on the placebo, that would not be ethical.”
For the record, having felt no side effects, I am fairly confident I received saline shots at my two Moderna study injection visits, not mRNA-1273. While I found it reassuring that Fauci and Moderna don’t expect me in the coming months to face the choice between abandoning the vaccine trial or receiving a vaccine from my healthcare provider, the decision ultimately resides at the FDA. Like tens of thousands of trial participants who suspect they aren’t vaccinated, I’m looking to the FDA to find us a reasonable pathway to vaccination that enables us to stay in the study.
Fortunately, potential solutions exist for current COVID-19 vaccine trial participants and future randomized control trials. These range from sharing data from a single control placebo control arm among numerous vaccine candidates, to competitive studies in which future vaccine candidates are tested against vaccines already approved, to emerging technologies that eliminate human placebo patients with synthetic or intelligent control arms and the “holy grail of correlates of protection.”
Early in my quest to understand the indecision surrounding placebo control arms, I reached out to Craig Lipset, a clinical trial expert and innovator who explained to me the world of pharmaceutical-grade placebo data. “The data from a pharma company control arm is clean, it is structured, and it is well organized,” Lipset tells me. It also contains no competitive intelligence about the vaccine being tested, so sharing control arm data among different sponsors can reduce the number of patients needed for a control arm. This summer, when vaccine makers were screening candidates to enroll in phase 3 vaccine trials, the head of therapeutics at Operation Warp Speed, Janet Woodcock, endorsed a cloud based platform, DataCelerate, designed “to share control arm data from ongoing and planned COVID-19 clinical studies.” The placebo database doesn’t replace control arms, it supplements them. Members in this not-for-profit initiative include Johnson & Johnson, Lilly, Pfizer, Merk, and Regeneron, to name a few.
A Digital Twin—an emerging technology not yet applied to infectious disease—uses machine learning to create an intelligent control arm populated with data from the active patient in a clinical study. The result is a “clinical record that describes what would have happened if a specific patient had received a placebo,” reducing the need for humans to receive placebos as part of the trial in question.
A third solution to the placebo paradox—and the holy grail of vaccine research—is called a “correlate of protection,” a measurement of a specific immune system marker that vaccine researchers could use to predict immunity requirements for protection from the virus that causes COVID-19, SARS-CoV-2. Last week, vaccine expert Dan Barouch and a team of researchers published their discoveries from a new primate study that suggests future vaccines could be licensed based on immune correlates, rather than clinical efficacy. Testing future Covid-19 vaccines swiftly and without human subjects removes the placebo control arm dilemma entirely and is already done annually with influenza vaccines.
To further these possible solutions, the 21st Century Cures Act was signed into law in 2016, mandating research into new ways of generating “real world evidence” from “sources other than randomized control studies.”
For today’s crisis, ensuring the safe approval of the quantities and variety of vaccines needed to supply the entire world will require the FDA and other regulatory bodies to consider implementing variants of these solutions, with the same open mind that allowed the initial rapid success of the Pfizer and Moderna vaccines. If the FDA finds a way for trial participants to make informed decisions and provides a reasonable endpoint for the studies, those of us who can safely and practically do so will not abandon our posts.
Together, we make the world safer.
The Bulletin elevates expert voices above the noise. But as an independent nonprofit organization, our operations depend on the support of readers like you. Help us continue to deliver quality journalism that holds leaders accountable. Your support of our work at any level is important. In return, we promise our coverage will be understandable, influential, vigilant, solution-oriented, and fair-minded. Together we can make a difference.
Keywords: Digital Twin, Moderna, Pfizer, correlate of protection, mRNA-1273 vaccine, placebo, randomized control trial
Topics: Biosecurity















I have been asked to participate in the Johnson & Johnson vaccine trial within the next few weeks and I have been thinking about this very issue. I was thinking that taking the antibody test would tell me whether I got the vaccine or the placebo. Am I missing something?
It seems recipients of the vaccine have low-grade fever, soreness and fatigue very soon after the dose. Thus the trial cannot be double-blind (neither researcher nor subject aware of assignment) and perhaps not even single-blind. “Correlate of protection” for future vaccines makes great sense.
My husband and I are volunteers in the Pfizer Phase 3 trial at Johns Hopkins. We hope to stick with the weekly reporting and occasional blood draws, COVID-19 tests through the two years. We are cancer survivors in our late 70s so represent a vulnerable group that we hope will be protected longer term than the 2 year study. Regardless if we receive the vaccine mid-study, we hope most volunteers stay scrutinized over time!
I am in the Johnson & Johnson study. I got my shot 3 weeks ago and have had zero side effects. My understanding is that the J&J shot (old school, not mRNA) has similar immediate side effects (or immune reaction) to the seasonal flu shot (which never gives me a reaction), so I don’t know if I received the vaccine or the placebo. I am not in any high risk group that is at the front of the line for covid vaccination, so I probably will not be able to get access to an approved vaccine until April or later.… Read more »
J &J has us agree NOT to take an antibody test once in the study. You get kicked out if they find out you had one. Also, they don’t tell you the results of the antibody tests they give you before and after the shot